
GW Pharmaceuticals’ cannabis-derived epilepsy drug wins European approval
The European Commission approved GW Pharma's Epidyolex as a treatment for seizures in two rare and severe forms of epilepsy. The drug won FDA approval last year.

The European Commission approved GW Pharma's Epidyolex as a treatment for seizures in two rare and severe forms of epilepsy. The drug won FDA approval last year.

The drug showed positive results in a 210-patient study in patients experiencing treatment-resistant seizures from tuberous sclerosis complex, a rare form of epilepsy.

How to turn analytics into actual policy outcomes.
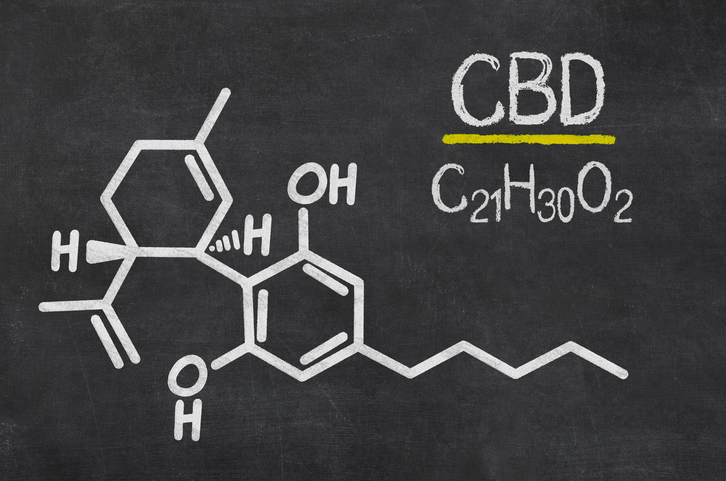
The FDA approved the cannabis-based drug for treating seizures in two severe forms of epilepsy.